

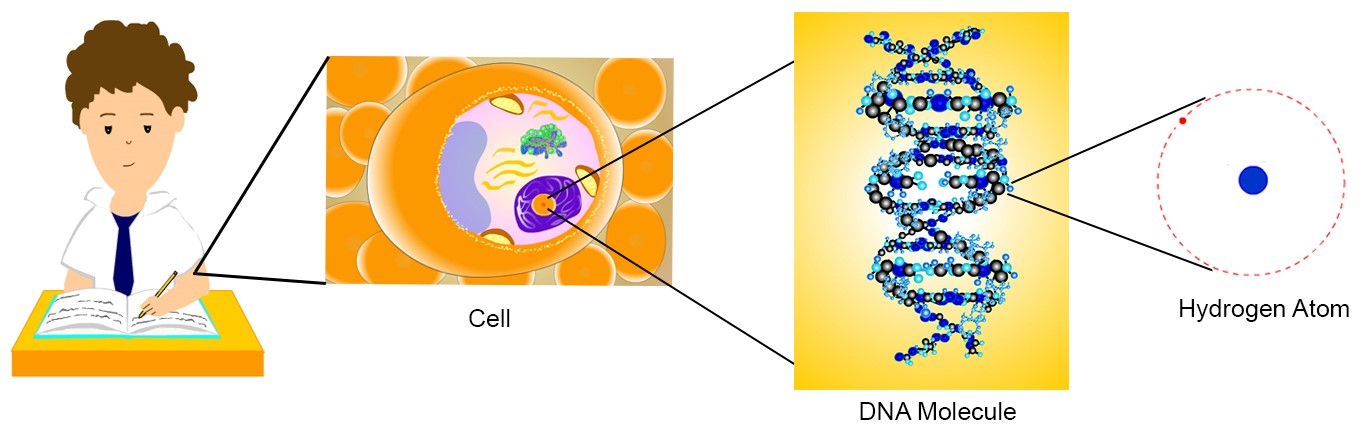
All matter—desks, books, clothes, and even your body, as well as all other animals and plants—is made up of atoms. Atoms are very tiny. For example, if you had a very powerful microscope, you could “zoom” in on one cell of your body. In the picture below, one cell in the arm of the boy is magnified (the second picture below). If you keep zooming in more and more, then you would be able to see DNA molecules that are in the cells. (This is shown in the third picture below.) The individual atoms that make DNA molecules are represented as the lighter and darker blue and black spheres. One of the atoms (hydrogen) that makes up a DNA molecule is represented on the far right.
Atoms are the smallest units of matter that can exist by themselves or in combination with other atoms. Smaller units of matter—such as electrons, protons, and neutrons—generally do not exist (for a very long time) on their own; they quickly combine with other particles that are nearby to form atoms! (By the end of this unit, you should have some idea why.)
Because of this, atoms are often called the “building blocks” of all matter. Atoms combine with other atoms to make molecules (including DNA), and more complex structures like chromosomes, cells, your body, and everything else in the universe!
Why should I care about atoms? Because atoms make up just about everything, it is very important to have a basic understanding of atoms. It's important to understand what atoms are made of and the forces that keep atoms together and let them bond with other atoms to make larger objects (including DNA molecules, different types of crystals such as salt and sugar, etc.).
Parts of atoms: protons, neutrons, and electrons. All atoms have a nucleus, which is the center of the atom. The nucleus of atoms is made of protons and neutrons. Atoms also have electrons, which are much smaller than protons or neutrons.
Electrons orbit—or move around—the nucleus at very high speeds. Two different kinds of atoms (or elements) are shown below. The atom on the left (hydrogen) has one proton and one electron. Hydrogen is the only type of atom/element that does not have any neutrons in its nucleus. The atom on the right (an oxygen atom) has 8 protons, 8 electrons, and 8 neutrons.
Important note: In the figure above, the circles around the atoms don't actually exist. The circles are called "Energy circles." These Energy circles represent about how much energy the electrons on that circle have (due to their motion and their potential for even more motion). The actual paths or orbitals of electrons in atoms can be very complex.
(For more information about Energy circles, you can click here.)
Charges of protons, neutrons, and electrons. A very important fact to remember is that these three different parts of atoms also have three different electric charges:
Electric force. Particles that have opposite charges (e- and p+) attract each other. Particles that have the same charge repel (or push each other away). This means that protons (p+), which are positively charged, repel each other. Also, electrons (e-), which are negatively charged, repel each other.
We call the force between charged particles the "electric force." The electric force is named after electrons.
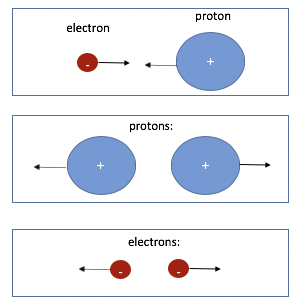
Because of the electric force, electrons and protons attract each other.
Because of the electric force, electrons repel (or push away) other electrons.
Finally, protons repel other protons because of the electric force.
Why the electric force is important. The electric force is very important to understand because it is responsible for atoms to even exist!! Our bodies (which are made of lots of carbon, oxygen, and hydrogen atoms) would not exist if it were not for the electric force! Trees and other plants would not exist! Animals would not exist! Planets would not exist! (You get the idea.)
The electric force is also responsible for a lot of things you may not have realized.

Strength of the Electric force. Let's say you have two electrons, which both have a negative charge. As the two electrons move farther from each other, the electric force pushing them away from each other gets smaller. This is shown below, where the length of the arrows represents the strength of the electric forces (Fe).
If you have two oppositely-charged particles (shown below), the electric force (Fe) pulling them toward each other increases as they get closer to each other.
In general, the strength of the electric force increases the closer the two charged particles are. And the strength of the electric force decreases as the charged particles move away from each other.
For more information about the strength of the electric force (Coulomb's Law), you can click here. (This is a bit more advanced: high school level.)
Masses of protons, neutrons, and electrons. Many people do not understand that all atoms—even the smallest ones that make up air—have some mass and weight! These masses are very small, and people might not be able to detect their mass, but they are made of matter. And all matter has mass! When you feel the wind (moving air) hitting your face, you can feel this because the atoms (and groups of atoms, called molecules) that make up the air have some mass.
The parts of atoms have masses (even though they are very very very small, from a human's point of view!).
The relative masses of protons, neutrons, and electrons are represented below (as a proportion of their volume):
The mass of an atom is the sum of the masses of its three parts: protons, neutrons, and electrons. So, if you know how many protons, neutrons, and electrons an atom has, you can figure out the mass of the atom. Or, you can just look up the mass of different types of atoms (called elements) in the Periodic table of elements.
When you're done with this game, Click the "Chemical & Physical Reactions" button above to see the list of units related to your research question.